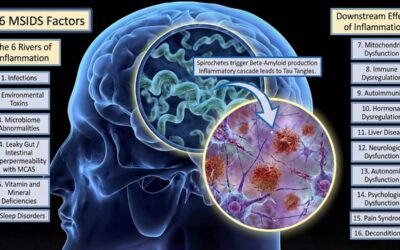
Alzheimer’s disease (AD) represents a significant medical concern for all of us as we grow older, as the NIH has published that the average risk of dementia over 55 years old is now 42%. AD is presently affecting 6.9 million Americans aged 65 or older and is expected...
Clinical Trials Roundup | 8 May 2026
Cytokinetics’ aficamten meets dual primary endpoints in the pivotal Phase 3 ACACIA-HCM trial for non-obstructive hypertrophic cardiomyopathy, Axsome Therapeutics secures FDA approval of AUVELITY as the first non-antipsychotic treatment for Alzheimer’s disease...
Paediatric Trials: Why Children Remain Therapeutic Orphans
Children have long been described as therapeutic orphans. It is a term first used by the American paediatrician Harry Shirkey of the Children’s Hospital in Birmingham, Alabama, at a 1963 conference and formally published in 1968. Despite more than five decades of...
Why Clinical Trial Diversity Remains a Problem
For decades, clinical trials have failed to adequately represent the patients who will ultimately use the medicines and treatments being tested. Gaps in enrolment across race, ethnicity, sex, age, and socioeconomic background have created an incomplete evidence base,...
Clinical Trials Roundup | 1 May 2026
Etavopivat delivers Phase 3 success in sickle cell disease, satralizumab becomes the first positive Phase 3 result in MOGAD, AstraZeneca completes its triple-positive COPD biologic programme, and a busy week for paediatric medicine brings new approvals and data in...
Speech Could be a Key Marker in Neurodegenerative Disease Drug Trials
Speech is rapidly emerging as an important, non-invasive, digital clinical marker in neurodegenerative disease research, according to leading biotech SynaptixBio. The company has facilitated a study run by Australia-based speech analytics company Redenlab, in...

Speech Could be a Key Marker in Neurodegenerative Disease Drug Trials
Speech is rapidly emerging as an important, non-invasive, digital clinical marker in neurodegenerative disease research, according to leading biotech SynaptixBio. The company has facilitated a study run by Australia-based speech analytics company Redenlab, in...

Most Supplement Companies Skip the RCT
Most Supplement Companies Skip the RCT. Here’s Why the Few That Don’t Often Get It Wrong The industry’s uncomfortable truth The global dietary supplement market is worth hundreds of billions of dollars.[1] Brands make claims about energy, cognition, immunity, and...

Clinical Trials Roundup | 24 April 2026
Neurona Therapeutics' rezanecel demonstrates durable seizure reduction past the one-year mark in drug-resistant mesial temporal lobe epilepsy at the AAN Annual Meeting, Gilead Sciences and Arcus Biosciences' TIGIT inhibitor domvanalimab fails its pivotal Phase 3 lung...

Panthera Biopartners Expands European Footprint
Panthera Biopartners Expands European Footprint with Acquisition of Hungary’s Leading Clinical Research Organisation, OEC Strategic acquisition marks the first step in Panthera’s European expansion, strengthening its global competitiveness Panthera Biopartners...

Clinical Trials Roundup | 17 April 2026
Revolution Medicines' daraxonrasib nearly doubles overall survival in previously treated metastatic pancreatic cancer in the Phase 3 RASolute 302 trial, Travere Therapeutics' Filspari receives FDA approval as the first-ever treatment for focal segmental...
Daraxonrasib Phase 3 Trial Results Signal a New Era for Pancreatic Cancer
The daraxonrasib pancreatic cancer trial results announced on 13 April 2026 represent what many in the oncology community are calling a landmark moment for one of medicine's most stubborn and lethal diseases. Revolution Medicines, a late-stage clinical oncology...

How Behavioral Science and Machine Learning Can Transform Trial Adherence
It’s ironic that the rigid nature of clinical research depends on a largely fragile variable: people. Even the most promising therapy cannot succeed in development if the study evaluating it struggles to keep participants engaged. Recruiting patients is already a...

Clinical Trials Roundup | 10 April 2026
Amgen’s subcutaneous reformulation of Tepezza meets its Phase 3 primary endpoint in thyroid eye disease, Lipocine’s oral brexanolone suffers a significant Phase 3 setback in postpartum depression, and NervGen Pharma secures FDA alignment on the RESTORE Phase 3...

Transforming Clinical Outsourcing: From Reactive to Predictive Strategy
The Growing Strain on Clinical Trial Operations Clinical trials have become increasingly diverse, complex, and expensive over the past decade. As the industry moves toward a more globalized model, managing timelines, coordinating across clinical functions, and...

Clinical Trials Roundup | 03 April 2026
Beam Therapeutics' base edited cell therapy for sickle cell disease earns a landmark NEJM publication, Immunovant's batoclimab misses in two Phase 3 thyroid eye disease trials, Seaport Therapeutics unlocks a novel anxiety treatment pathway, and Eli Lilly's oral GLP-1...

International Menopause Society Unveils Global Care Framework
International Menopause Society sets new global standards for menopause care with evidence-led framework For the first time in a decade, the International Menopause Society (IMS) has released a complete redevelopment of its global menopause recommendations. Grounded...

Clinical Trials Roundup | 27 March 2026
An RNA therapy that preserves muscle while cutting visceral fat, a first in class oral non incretin obesity pill, late stage lung cancer portfolio data, NK cell immunotherapy in Alzheimer’s disease, and the first human trial of a cellular rejuvenation medicine...