Not long ago, I spoke with a colleague, a mother named Kelley who has spent the past two years asking a question that increasingly reflects a broader challenge in oncology:
If technologies exist to detect cancer earlier, why aren’t we dedicating more effort to support them?
Her daughter, Rachel, was 26 when she was diagnosed with Ewing sarcoma, a rare and aggressive malignancy. Rachel underwent chemotherapy and radiation, and at one point achieved what imaging suggested was a complete response. However, within months, the cancer recurred.
Only after Rachel’s death did Kelley learn about emerging diagnostic approaches—particularly minimal residual disease (MRD) and circulating tumor DNA (ctDNA) testing. In several tumor types, these modalities have demonstrated the ability to detect molecular recurrence months in advance of radiographic progression.¹
She asked me: Why didn’t I know about these tests? And could they have seen what the imaging didn’t?
Earlier detection may not have altered Rachel’s outcome. However, this question is increasingly relevant at a systems level: whether earlier molecular insight could have informed different clinical decisions, including surveillance strategies or therapeutic intervention.
In oncology, where outcomes are highly stage-dependent, this question is not theoretical. Five-year survival rates for many cancers exceed 90% when detected at localized stages but decline significantly in metastatic disease.²
The Gap Between Innovation and Clinical Integration
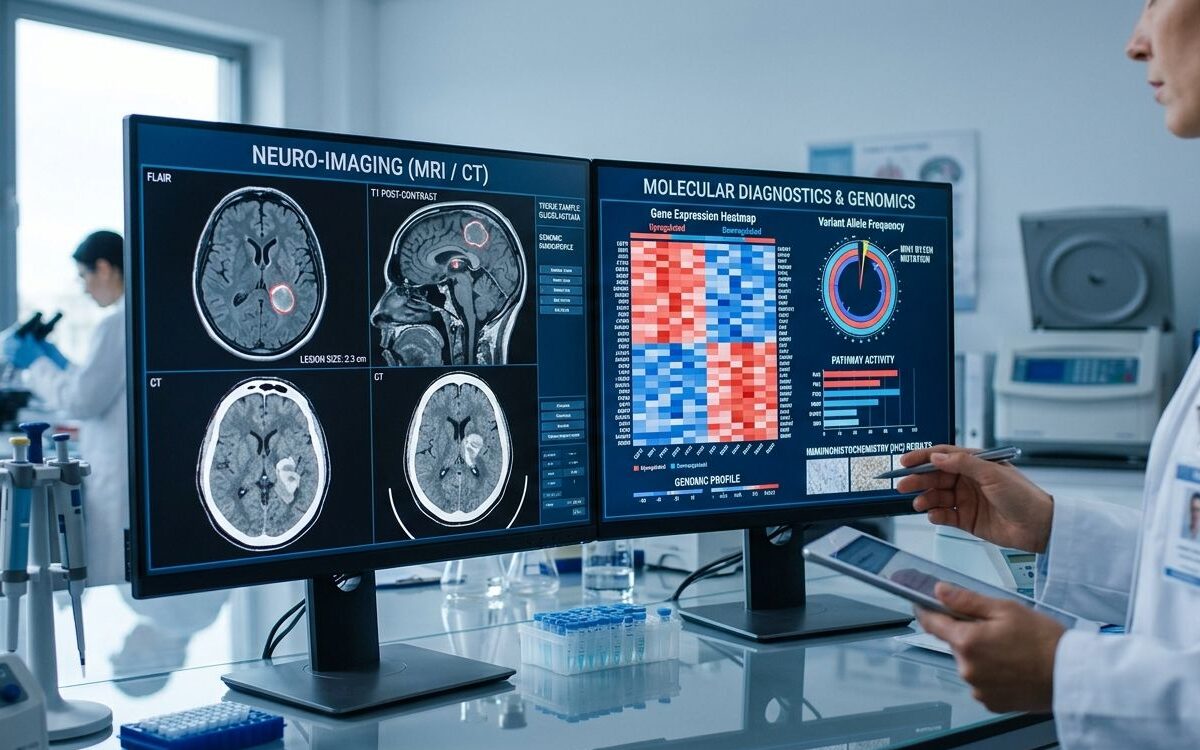
Advances in oncology diagnostics over the past decade have been substantial. MRD and ctDNA assays are demonstrating clinical utility across multiple indications, including colorectal, lung, and hematologic malignancies, particularly in detecting recurrence and monitoring treatment response.³
In parallel, multi-cancer early detection (MCED) technologies are progressing through large-scale validation studies, with the potential to identify malignancies prior to symptomatic presentation.⁴
Of course, more research is required to truly validate these tests, but an absence of evidence does not equal evidence of failure.
And we need to be looking at the appropriate endpoints for the appropriate patients. Perhaps it’s too soon for regular, precautionary testing in every patient. But for patients with active cancer, or those in remission, these molecular assessments may have a truly meaningful impact on treatment plans.

Rebalancing the Oncology Paradigm
The oncology ecosystem has historically directed the majority of investment toward therapeutics, particularly for advanced disease.
In the United States alone, oncology drug spending exceeded $100 billion annually in recent years, while investment in early detection and diagnostics remains comparatively limited.⁵ At the same time, late-stage cancer care is associated with substantially higher costs and poorer outcomes relative to earlier-stage intervention.⁶
These contradictions underscore a fundamental challenge: therapeutic intervention alone cannot compensate for delayed detection.
Diagnostics are not ancillary to oncology care; they are foundational. Earlier detection of recurrence, real-time monitoring of treatment response, and improved risk stratification have the potential to significantly alter both clinical outcomes and cost trajectories.
The Infrastructure Enabling Diagnostic Innovation
Less visible, but equally critical, is the infrastructure required to support diagnostic development.
The validation of emerging diagnostics depends on access to high-quality biospecimens, longitudinal data, and rigorously annotated clinical datasets. Variability in specimen quality or data integrity can delay validation timelines, introduce inconsistencies in results, and ultimately impede regulatory and clinical adoption.
These upstream dependencies are often underrepresented in broader discussions but play a central role in determining how efficiently new diagnostics progress from research to clinical use.
In a time-sensitive disease context, these delays are not trivial. The pace at which diagnostics can be developed, validated, and implemented has direct implications for patient outcomes.
An Increasingly Informed Patient Population
At the same time, the patient landscape is evolving. Access to medical information has expanded significantly, and the emergence of AI-enabled tools is further accelerating this trend. Patients and caregivers can now interpret clinical reports, review relevant literature, and generate informed questions with a level of autonomy that was not previously possible.
This shift is contributing to a more engaged patient population—one that is increasingly aware of diagnostic advancements, even when those tools are not yet part of standard care pathways.
However, this growing awareness has also exposed inconsistencies in access and adoption. Even well-informed patients may encounter limitations related to test availability, physician familiarity, reimbursement, or guideline inclusion.
This dynamic is creating a widening gap between what is scientifically feasible and what is operationally delivered in routine care.
Aligning Innovation with Implementation
It’s clear that innovation, awareness, and implementation are not yet fully aligned.
The field has made significant progress in developing tools capable of detecting cancer earlier and more precisely. The remaining challenge is ensuring that these tools are integrated into care pathways in a timely, evidence-based manner.
Addressing this gap will require coordinated action across stakeholders:
- Accelerating the incorporation of validated diagnostics into clinical guidelines
- Expanding physician education and decision-support resources
- Improving reimbursement frameworks to support appropriate utilization
- Enhancing patient access to clear, evidence-based information
- Strengthening the research infrastructure that underpins diagnostic development
The question people like my colleague Kelley are asking—whether earlier detection would have made a difference or saved a loved one’s life—is increasingly one the industry must be prepared to answer.
As diagnostic capabilities continue to advance, the focus must shift from innovation alone to implementation at scale. Because in oncology, time is not simply a variable. It is a determinant.
Author Bio

Inga Rose, Founder of Reference Medicine
Inga Rose is the Founder of Reference Medicine, and has spent her career serving the healthcare industry, primarily focused on oncology clinical research and diagnostics. Reference Medicine offers a comprehensive product line of specimens tailor-made for oncology test developers at a fraction of the cost – giving patients meaning to their disease and helping biotech teams spend less time sourcing and more time building the next life-saving diagnostic.