Kelly Nguyen is a molecular biologist and Group Leader in the Structural Studies Division at the MRC Laboratory of Molecular Biology (LMB), where her research aims to uncover how telomeres are structured, maintained and regulated by the enzyme telomerase.
Telomeres are protective nucleoprotein complexes that cap the ends of eukaryotic chromosomes and consist of repetitive, G-rich DNA sequences (TTAGGG in humans). They play a critical role in preserving genome stability and ensuring cell viability. Telomere length is controlled by telomerase, a ribonucleoprotein enzyme that extends the 3′ ends of chromosomes by adding telomeric repeats. These structures are critical to our understanding of human health as loss of telomeres is linked to ageing, while aberrant telomerase activation is a hallmark of many cancers.
Kelly first began researching telomerase in 2016 as a Miller Fellow postdoc at the University of California, Berkeley with Kathleen Collins and Eva Nogales. She launched her own research group at the LMB in 2019.
At the LMB, Kelly’s group focuses on how cells overcome two key challenges in telomere biology: compensating for telomere shortening caused by incomplete DNA replication, and protecting chromosome ends from being erroneously detected as DNA breaks. In parallel, the team seeks to elucidate the structure and function of telomerase, with the goal of understanding how changes in its activity influence telomere maintenance.
To investigate telomere biology, Kelly’s group uses a combination of biochemistry, structural biology and functional in-cell studies. This approach has led to several breakthroughs already, including the determination of the first ever atomic model of human telomerase. The telomerase was prepared by extracting it from cultured human cells before imaging using electron cryomicroscopy (cryo-EM). The resulting data was analysed using RELION, a computer programme developed by Sjors Scheres’ group at the LMB, resulting in a high-resolution structure of the human telomerase which revealed a previously unknown histone dimer as a telomerase subunit and a hotspot of premature ageing disease mutations.
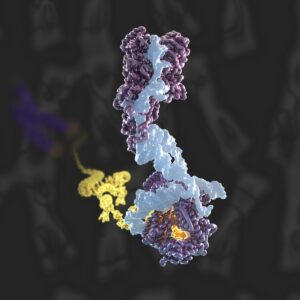
Structure of human telomerase bound to the chromosome ends for telomeric DNA extension (Nguyen et al., Nature, 2018; Ghanim et al., Nature, 2021).
Additionally, Kelly’s group’s work with cryo-EM has shown how part of the protein complex shelterin recruits and activates telomerase to extend telomere ends and modulates telomeric nucleosomes. These findings lay the groundwork for understanding how shelterin and nucleosomes, together with telomerase, contribute to the formation of telomeric caps at chromosome ends, a necessary step in preventing DNA damage.
More recently, Kelly’s group has continued to deliver exciting and impactful publications, including the first structure of human telomerase in a dimeric formation. In collaboration with Yiliang Ding’s group at the John Innes Centre and Rhiju Das’s group at Stanford University, the group confirmed the existence of a dimeric state of human telomerase. Disruption of the dimerization interface resulted in compromised telomerase assembly and shortened telomeres in cells. Importantly, the dimerization interface site was found to contain a cluster of several mutations linked to premature ageing disorders, providing a potential explanation for why telomere maintenance is affected in some ageing diseases.
Overall, through detailed investigation of telomere structure and telomerase regulation, Kelly’s research carries strong clinical relevance. Understanding how telomeres are maintained and shortened offers valuable insight into ageing and age-associated diseases, while studies of telomerase activation contribute to our knowledge of cancer development. Collectively, her group’s work may pave the way for new therapeutic approaches.
On what sparked her interest in studying telomere biology and the future possibilities, Kelly commented:
“Despite representing only a small fraction of our genome, telomeres have a profound influence on aging, cancer development and the regenerative capacity of tissues. By visualising telomere-related processes in molecular detail, we can directly link structure to biological function and disease mechanisms. We hope that the knowledge generated through this work will ultimately contribute to more effective therapeutic interventions in the future.”
Kelly’s contributions to the field have been recognised by several awards including the prestigious 2025 Lister Research Prize, the 2024 Colworth Medal from the Biochemical Society, the 2022 Eppendorf Award for Young European Investigators, the 2020 Suffrage Science Award curated by the MRC London Institute of Medical Sciences (LMS) and the 2017 Early Career Research Award from the Biochemical Society. Most recently, she was honoured as Laureate for Life Sciences at the 2026 Blavatnik Awards in the UK on the 24 February 2026.
Kelly reflected that she is thankful to the members of her group, colleagues at the LMB, and mentors and collaborators for their continued support throughout her research career, commenting:
“Science is truly a team endeavour. Although the path can at times be challenging, the knowledge that our discoveries may one day contribute to improving human health continually motivates us to push boundaries and strive further.”