As the echoes of the COVID-19 crisis fade, humanity remains on high alert for the inevitable next pathogen. What if we could dissect viruses down to their atomic core, revealing chinks in their armor that lead to revolutionary treatments? Drawing from my 12+ years as a structural biologist—from pioneering enzyme structures during my Ph.D. at the Indian Institute of Technology Bombay to advancing HIV research at Meharry Medical College in Nashville—I’ve witnessed how these microscopic revelations drive real-world impact. This piece uncovers cutting-edge advances in combating HIV, SARS-CoV-2, and the high-risk Nipah virus, showcasing structural biology’s role in not just treating, but preempting pandemics through innovative drug strategies.
HIV’s Stealthy Assault: Atomic Revelations Paving the Way for Eradication
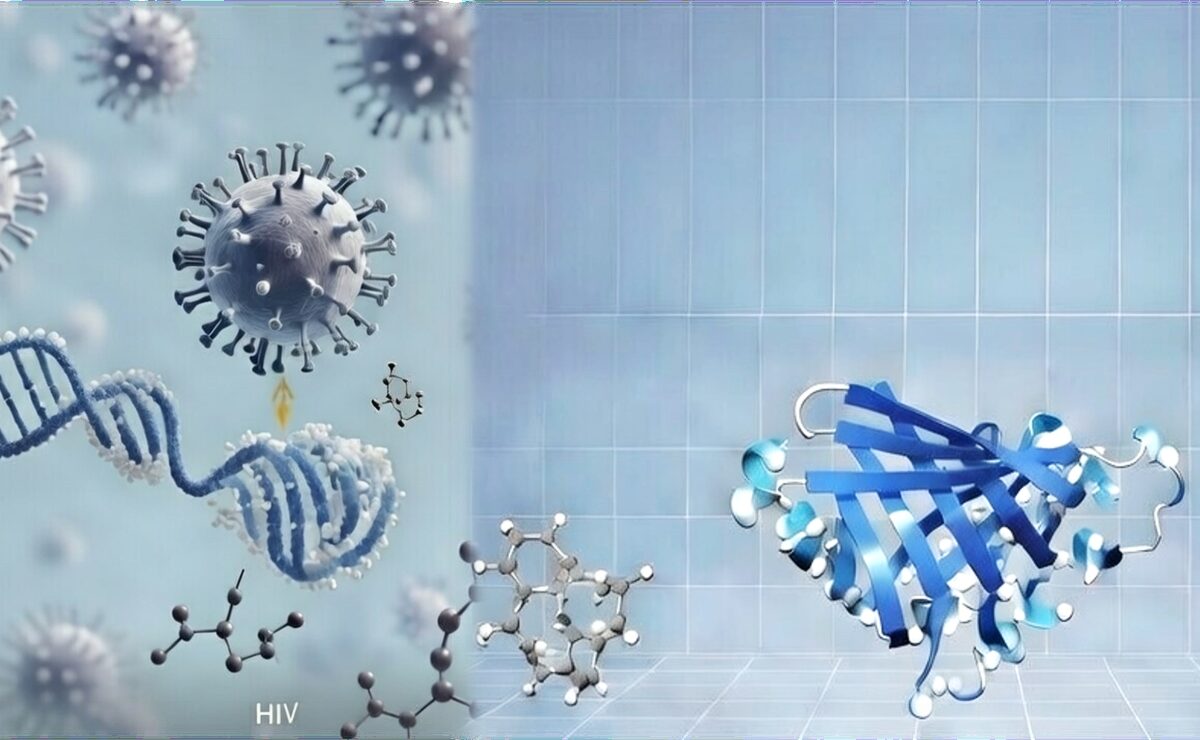
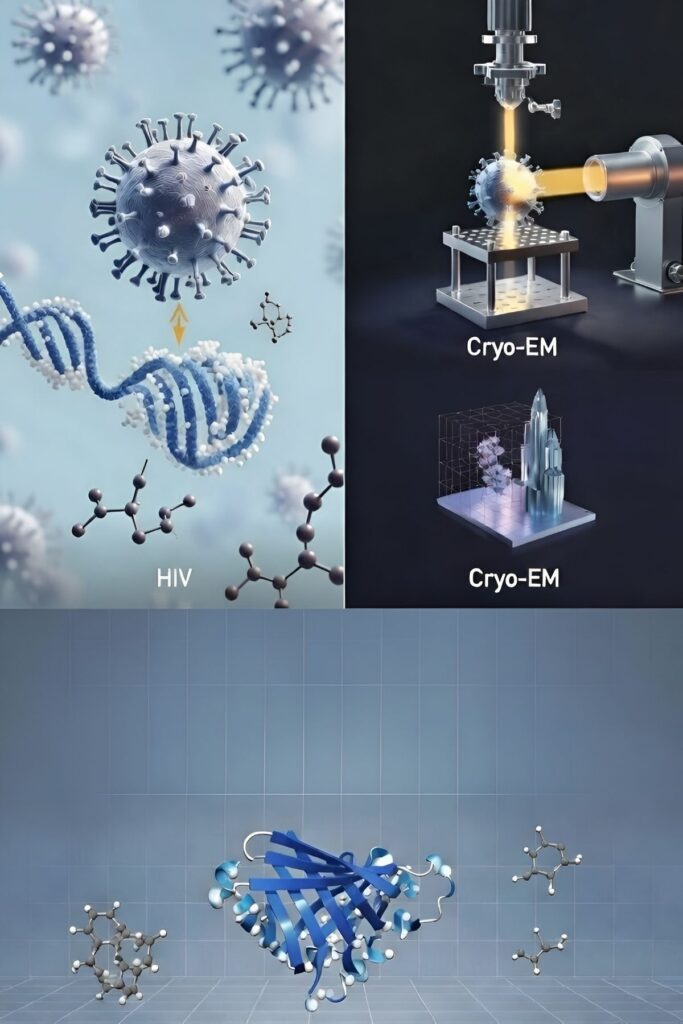
At the heart of HIV’s tenacity is its integration into human DNA, forming reservoirs that evade even the best antiretrovirals. Cutting-edge structural analyses are now exposing vulnerabilities here. My team’s 2024 research in the Journal of Biological Chemistry employed X-ray crystallography, molecular dynamics, and fluorescence techniques to demonstrate how the host enzyme TREX1 selectively dismantles faulty HIV DNA, thwarting immune evasion. Visualize HIV as a cunning spy embedding secrets in a fortress—TREX1 serves as the demolition expert targeting weak links, though the virus often slips through. This breakthrough, recognized with the Daniel Wolf Prize, opens doors to drugs that enhance TREX1’s precision, potentially activating innate immunity via pathways like cGAS to diminish viral hideouts.
Echoing this, a 2025 review in the Journal of General Virology references our data to illustrate the capsid’s pivotal role in dodging host defenses, fueling infection spread. Structural models from cryo-electron microscopy portray the capsid as a geodesic dome of protein subunits; disrupting its assembly, as with Gilead’s lenacapavir—hailed as Science’s 2024 Breakthrough—offers prolonged protection, up to six months in clinical trials.
Complications arise from comorbidities such as cardiovascular issues. In my lead-authored 2024 piece in Circulation Research, we dissected mechanisms like persistent inflammation and vascular damage in HIV patients, calling for integrated therapies. Projections in a 2024 Lancet HIV study extend this to 2050, underscoring the urgency for antivirals that address both infection and secondary health burdens. Recent CROI 2026 updates on combinations like bictegravir and lenacapavir highlight 96-week viral suppression, all anchored in these atomic-level discoveries.
Broadening the Fight: Structural Tactics Against SARS-CoV-2 and Nipah’s Menace
The power of structural biology transcends HIV, offering blueprints for other viral foes. For SARS-CoV-2, rapid cryo-EM mapping of the spike protein accelerated mRNA vaccines. Emerging from a 2025 multi-institute effort, the compound ASAP-0017445 targets the main protease across variants, showing robust preclinical profiles. Innovations like phage-displayed nanobodies from the Midwest Antiviral Drug Discovery Center adapt to mutations, blocking entry in Omicron strains.
Then there’s Nipah, a zoonotic killer with fatality rates nearing 70%, poised for wider outbreaks. In my February 2026 Medical News Today op-ed, I spotlighted its bat-to-human jumps and the critical need for proactive countermeasures. A 2024 study introduced the monoclonal antibody hu1F5, which neutralizes the fusion glycoprotein in primate models. Biophysical simulations in 2025 pinpointed procyanidins as potent inhibitors binding Nipah’s G protein through adaptive fitting. These strategies draw parallels to my doctoral breakthroughs on glutamate dehydrogenase (GDH), where I crystallized 10 novel complexes to decode metabolic regulation—insights now transferable to how viruses like Ebola or Zika hijack cellular pathways, inspiring metabolism-targeted antivirals.
Hurdles in the Hunt: Strategies to Sidestep Common Traps in Antiviral Development
Progress isn’t without pitfalls. Viral diversity can render drugs obsolete quickly, akin to the limitations seen in initial KRAS oncology inhibitors that overlooked mutational breadth. Relying solely on frozen structures ignores fluid interactions—hence, my advocacy for blending simulations with empirical data, as in our TREX1 investigations.
Proven approaches include upfront biophysical screenings (e.g., surface plasmon resonance for affinity) alongside CRISPR-based functional tests. Adhering to regulatory frameworks like ICH Q11 ensures viable paths to approval, while incorporating pharmacogenomics—per CPIC standards for HIV regimens—tailors treatments to mitigate side effects like those exacerbating hypertension.
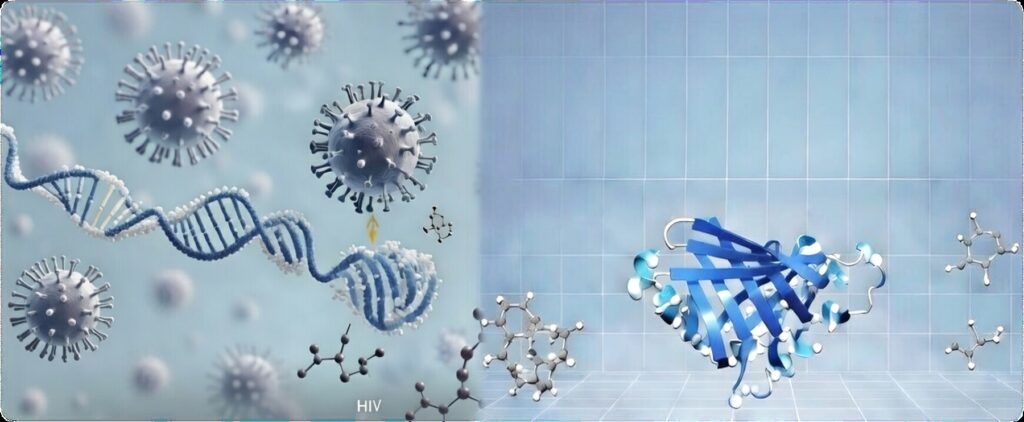
Artistic representation of viral molecular integration into host DNA, visualized through structural biology techniques. On the left, the virus interacts with a DNA helix amid surrounding molecules, illustrating atomic-level evasion tactics. On the right, a ribbon model of a viral protein target, flanked by small-molecule inhibitors, demonstrates potential sites for antiviral drug design. This image encapsulates how structural biology reveals vulnerabilities in HIV and similar pathogens, paving the way for next-generation therapies against current and emerging pandemics.
Future Frontiers: Harnessing AI and Novel Modalities for Universal Protection
Artificial intelligence is reshaping the landscape: Tools like AlphaFold predict viral architectures swiftly, expediting inhibitor design. My explorations of chromatin complexes suggest epigenetic agents as future disruptors; meanwhile, PROTACs and tri-complex systems tackle previously intractable proteins. The 2025 INTREPID Alliance pipeline overview catalogs over 100 preclinical candidates, with SARS-CoV-2 leads informing cross-viral applications.
Looking ahead, imagine cryo-EM and AI converging to destabilize viral envelopes, prompting infected cells to self-eliminate. Initiatives like the COVID Moonshot exemplify open collaboration for accessible, patent-free solutions. Informed by my GDH and TREX1 endeavors, this fusion promises equitable therapies that combat infections, associated conditions (e.g., cardiovascular risks in HIV), and emerging threats holistically.
Forging Alliances: Your Role in a Pandemic-Resistant Era
Structural biology isn’t confined to labs—it’s a collective arsenal decoding viral enigmas from HIV’s integration tricks to Nipah’s surface proteins. As an educator at Meharry, I champion mentorship to amplify diverse voices in this fight. Engage by supporting research advocacy, exploring resources like AlphaFold tutorials, or connecting via platforms like INTREPID. Let’s collaborate to neutralize future pandemics—reach out, and together we’ll innovate for a healthier world.
Author Bio:

Prem Prakash, Ph.D.
Prem Prakash, Ph.D., serves as a Research Scientist at the Center for AIDS Health Disparities Research, Meharry Medical College, Nashville, TN-37208. An expert in protein crystallography, enzymology, and antiviral drug design, he has over 20 publications and actively reviews for top journals.
Google Scholar: https://scholar.google.com/citations?user=Uk6TEwgAAAAJ&hl=en
LinkedIn: https://www.linkedin.com/in/prem-prakash-ph-d-779160160/
ResearchGate: https://www.researchgate.net/profile/Prem-Prakash-15