The Growing Strain on Clinical Trial Operations
Clinical trials have become increasingly diverse, complex, and expensive over the past decade. As the industry moves toward a more globalized model, managing timelines, coordinating across clinical functions, and maintaining quality have all become critical to delivering a well-executed study. Research conducted by Markey et al. (2024) confirms that clinical trial complexity—measured through extracted trial data such as endpoints and inclusion-exclusion criteria—has risen not only across development phases but also across therapeutic areas. The authors attribute this growing complexity to a greater burden on patients and investigators, rising costs, and longer timelines to bring medicines to market (Markey et al., 2024).
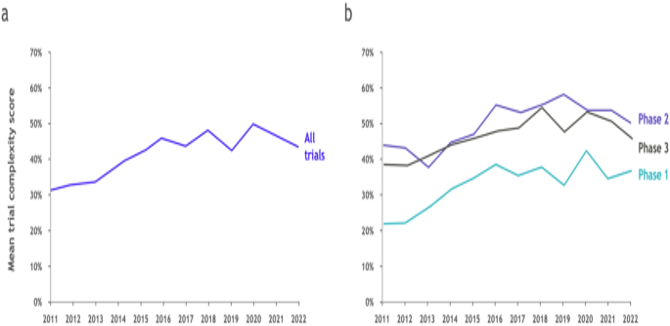
Figure 1: The diagram shows the evolution of mean trial complexity across the 3 phases (Markey et al., 2024)
Within this landscape, clinical outsourcing plays a strategically important yet often overlooked role in trial execution. Outsourcing teams are not only responsible for managing trial timelines—ensuring that third-party vendors and sponsor clinical operations teams have sufficient start-up time—but also for driving cost efficiency by negotiating competitive rates without compromising service quality.
Historically, however, outsourcing strategies have been reactive, manual, and fragmented. Organizations have tended to rely on static preferred vendor lists, evaluate bids through largely manual processes, and approach negotiations with an emphasis on cost reduction rather than overall value. As Cooper (2024) observes, the pressures of globalization, technological advancement, and shifting market dynamics now demand a more sophisticated and strategic approach to vendor selection. The question is no longer whether outsourcing models need to evolve, but how quickly the industry can make that shift.
The Current State: Reactive and Fragmented Outsourcing Models
At present, vendor selection in clinical outsourcing relies heavily on previously developed and approved vendor lists. While the rationale behind maintaining such lists is understandable—enabling organizations to assess a vendor’s quality systems, security protocols, and data privacy practices—the process of onboarding a new vendor remains arduous. This often dissuades teams from engaging with newer vendors who might bring fresh capabilities to a clinical trial sooner rather than later.
This challenge is compounded by what Kern, Willcocks, and Heck (2002) describe as the “winner’s curse,” a phenomenon in which vendors undercut their prices to secure a contract, sometimes making irrational bidding promises and potentially finding themselves unable to recover their business and operating expenses once the work begins (Adobor, 2012).
Contracting and budget negotiations present another significant bottleneck. Prolonged discussions can delay study start timelines considerably, often because organizations lack standardized benchmarks against which to compare proposals. Even where rate cards exist, they may not be sufficiently tailored to accommodate the specific requirements of different clinical trials.
Beyond the financial dimension, outsourcing clinical trials to a third party such as a contract research organization (CRO) inevitably involves some loss of direct control. This can result in misaligned priorities or breakdowns in communication between the sponsor and the CRO. In an article by Getz (2018), a Tufts CSDD survey of 88 pharmaceutical and biotechnology companies found that sponsors report mixed levels of satisfaction with both their outsourcing management capabilities and their oversight effectiveness. Companies were most satisfied with oversight reporting accuracy but least satisfied with the clarity and practicality of proposed resolutions. Faster issue identification and resolution, along with more actionable insights and recommendations, were identified as the top areas where oversight could most improve. The article also notes that the application of tools and technologies would go far in helping to enable better communication, reporting, and accountability—a finding that directly points toward the need for the kind of data-driven solutions explored in the next section.
Taken together, these challenges—from rigid vendor lists and the winner’s curse to weak benchmarking and limited oversight effectiveness—paint a clear picture of an outsourcing function in need of modernization. Reducing start-up timelines and improving operational alignment are not merely matters of efficiency; they are integral to saving time, money, and ultimately, patient lives. What the industry needs is a model that replaces reactive decision-making with predictive intelligence—and that model is beginning to take shape.
The Shift to Predictive Outsourcing: A New Operating Model
The growing availability of artificial intelligence presents an opportunity to move beyond these entrenched limitations and toward predictive, data-driven outsourcing models. This new paradigm integrates predictive analytics and AI tools with CRO partnerships, enabling organizations to forecast needs, optimize resource allocation, and manage trial operations proactively—both before and during execution.
Crucially, these tools can address several of the structural weaknesses outlined above. Based on research by Rainy et al. (2022), automating adaptive performance scorecards and utilizing algorithms like Support Vector Machines (SVM) allows AI to flag “silent risks” and unrealistic performance indicators. This continuous, holistic oversight ensures that issues like non-compliance are detected early, significantly improving procurement agility and supply chain resilience. Support Vector Machines can be used to create a buffer zone that accurately classifies vendors as reliable versus risky based on historical data, rather than penalizing vendors over minor fluctuations. If adapted for clinical vendor evaluation, this approach would support data-driven decisions rather than relying on intuition alone—offering a practical countermeasure to the winner’s curse by enabling sponsors to identify unrealistically low bids before awarding a contract. Similarly, AI-driven benchmarking tools trained on historical pricing and performance data across therapeutic areas, geographies, and trial phases could generate dynamic, study-specific benchmarks, giving negotiators something concrete to compare bids against rather than relying on static or outdated rate cards.
AI is already demonstrating its value in clinical trial design and patient recruitment through predictive analysis (Saini et al., 2025). It can help determine optimal sample sizes and trial durations, often enabling “leaner” trials that require fewer participants while preserving statistical power. AI-driven designs also support the creation of adaptive trials, in which protocols can be adjusted in real time based on interim data analysis (Saini et al., 2025). As these capabilities mature, it becomes increasingly important for pharmaceutical companies to partner with third-party vendors who can leverage such technology to deliver data-driven solutions efficiently. This in turn improves oversight for outsourced trials and enables better communication through the immediate availability of data.
While the manual review of requests for proposals (RFPs) and requests for information (RFIs) will remain necessary in the initial stages, onboarding vendors who utilize AI-based systems can help sponsors make faster, more data-informed decisions. According to Saini et al. (2025), AI-based systems can also assist in predicting legal and financial risks associated with vendor engagements. Several software platforms already on the market can review RFPs based on historical data and integrating with platforms such as Google Drive and OneDrive. Research by Roshan et al. (2025) demonstrated the effectiveness of an AI-assisted tender evaluation system, in which eligibility criteria—including experience from similar assignments and financial capability thresholds—were provided as inputs. The study reported a 60% reduction in bid evaluation time, with the AI system achieving accuracy rates exceeding 90%. Depending on organizational requirements, such tools can be developed in-house or outsourced, and they hold promise for making bid defenses more efficient and negotiations faster.
The overarching benefit of adopting AI in outsourcing—whether through vendor partnerships or internal development—is that it acts as a decision-support tool, helping to compress clinical trial timelines. Importantly, throughout the use of these systems, a human-in-the-loop remains essential to verify data accuracy and make the necessary judgment calls.
Practical Challenges to Implementation
Despite its promise, the adoption of AI in clinical outsourcing is not without obstacles—and overlooking them would be a mistake. Research by Afifa et al. (2025) identifies human and organizational factors as the most significant barriers to adoption, often exerting a greater influence than financial or technological considerations. A primary obstacle is deep-seated resistance to cultural change among experienced staff, alongside limited engagement from middle management. For organizations accustomed to established ways of working, the introduction of AI tools can feel more like a disruption than an improvement, particularly when the benefits are not yet tangible.
Data privacy and algorithmic bias present additional barriers. As Pesqueira et al. (2024) note, security concerns center on protecting sensitive commercial data and ensuring the robustness of systems against cyber threats, given that breaches could compromise competitive bidding strategies. Bias is also a critical risk: when AI models are trained on historical data that reflects past human prejudices or systemic inequalities, they may produce unfair pricing or discriminatory outcomes in tender evaluations. Addressing these issues will require a combination of robust governance frameworks, transparent data practices, and ongoing human oversight.
There is also the matter of cost. Implementing AI-driven solutions requires significant upfront investment, not only in technology and software but also in the personnel needed to operate, maintain, and optimize these systems (Vellanki, 2024). For smaller biotech firms with limited budgets, these financial hurdles can make effective AI adoption particularly difficult.
Finally, the regulatory landscape has yet to catch up. AI is evolving rapidly, but standardized guidelines for GxP compliance remain limited. At present, many life sciences companies lack clear frameworks to assess and validate AI solutions, which creates uncertainty about how to adopt the technology responsibly across the organization (Vellanki, 2024).
Conclusion: Rethinking Outsourcing as a Strategic Function
The clinical trial landscape is evolving at a pace that traditional outsourcing models can no longer keep up with. As trial complexity increases and the pressure to reduce development timelines intensifies, the industry must move beyond reactive procurement and embrace outsourcing as a strategic function. The problems are well documented: rigid vendor lists that stifle competition, a winner’s curse dynamic that incentivizes unsustainable bids, weak benchmarking that leaves negotiators without reliable reference points, and oversight structures that remain reactive rather than proactive. These are not minor inefficiencies—they are systemic issues that directly impact how quickly and cost-effectively new therapies reach patients.
The integration of artificial intelligence and predictive analytics offers a credible pathway toward addressing these challenges. Tools like AI-powered performance scorecards and automated tender evaluation systems are already demonstrating measurable gains in speed and accuracy, while predictive models offer the potential to flag unrealistic bids, generate dynamic benchmarks, and streamline vendor qualification. At the same time, AI-enabled trial design and patient recruitment capabilities are creating new expectations for what CRO partnerships can deliver. However, realizing this vision will not be straightforward. Organizations must confront the cultural resistance that often accompanies technological change, invest in data governance to mitigate bias and privacy risks, navigate an evolving regulatory environment for GxP compliance, and secure the financial resources needed to implement these systems—a challenge that falls disproportionately on smaller firms. The shift from a cost-driven to a value-driven outsourcing model requires not only new tools but also a fundamental change in mindset—one that positions outsourcing professionals as strategic partners in clinical development rather than administrative gatekeepers. For pharmaceutical and biotechnology companies willing to make this transition, the rewards are significant: shorter start-up timelines, stronger vendor relationships, improved regulatory readiness, and ultimately, a faster path to delivering life-changing therapies to the patients who need them.
Author Bio
Aishwarya is a clinical outsourcing professional with over six years of experience in the healthcare and pharmaceutical industries. Specializing in vendor negotiations, contract strategy, and process optimization, Aishwarya has worked across clinical trials of varying scope and complexity, while ensuring compliance with legal and regulatory standards. With a strong foundation in analytical problem-solving and stakeholder collaboration, Aishwarya writes about the intersection of outsourcing strategy, emerging technology, and clinical trial operations.
Disclaimer: The author works in strategic outsourcing and vendor management within the pharmaceutical industry. This article reflects her own independent analysis and is provided for informational purposes only; it does not constitute professional advice from Life Science Daily News.














