At the 2026 Congress of the European Society of Gynaecological Oncology (ESGO) in Copenhagen, Dr Gabriel Levin of the McGill University Health Centre will present Phase II data evaluating the investigational DNA therapy Elenagen in combination with gemcitabine for platinum-resistant ovarian cancer (PROC); one of the most aggressive and treatment-refractory settings in gynecologic oncology.
The randomized study, recently published in the International Journal of Gynecological Cancer, reported a doubling of median overall survival, from approximately 13 months with chemotherapy alone to more than 25 months with the addition of Elenagen. The combination also reduced the risk of death by nearly 60%, without an observed increase in treatment-related toxicity.
While the survival figures are striking, the biological and practical implications of the findings may prove just as significant as the headline numbers.
The “Legacy Effect”: Insights from an Unplanned Interruption
One of the most unusual aspects of the trial was a non-medical interruption in drug supply that required all patients to discontinue Elenagen simultaneously, regardless of how long they had been receiving treatment.
Dr Vlad Gabai, Vice President of R&D at CureLab Oncology, emphasised that these interruptions were not linked to safety concerns.
“First and foremost, it must be articulated that no unplanned treatment interruptions were attributed to treatment-emergent adverse events, which is typically the cause in oncology trials. Instead, these synchronous interruptions occurred due to non-medical logistical reasons.”
The unexpected pause created an observational window. According to Gabai, during the first 12 months of therapy, longer exposure to Elenagen “strongly correlated with longer patient survival after the treatment was stopped.”
However, the incremental benefit appeared to taper beyond that period.
“The additional benefit of Elenagen appeared to diminish between the 12th and 24th months. Thus, although statistically positive, the extension of patient survival beyond 18 months of treatment was relatively minimal.”
This observation is shaping future trial planning. Gabai notes that upcoming U.S. and EU protocols will likely assume treatment durations of approximately 18–24 months, unless patients elect to discontinue.
He also stressed caution regarding mechanistic interpretation.
“There have been attempts to interpret this observation as indirect proof of CureLab’s mechanism of action hypothesis, specifically, that Elenagen remodels the tumor microenvironment to enhance the anti-cancer immune response. However, these conclusions are premature.”
Definitive confirmation would require serial tumor biopsies during and after treatment; procedures not performed in the trial for ethical and legal reasons.
“This valuable clinical observation cannot be used as definitive mechanism-of-action proof at this stage. CureLab maintains a rigorous commitment to evidence-based claims, avoiding speculative conclusions without direct confirmation.”
The CA-125 Signal: Why Higher-Risk Patients Responded More Strongly
The study also generated interest around patients with elevated CA-125 levels, traditionally associated with higher tumor burden and poorer prognosis.
Counterintuitively, these patients appeared to derive disproportionate benefit from the Elenagen combination.
Gabai offered several potential biological explanations.
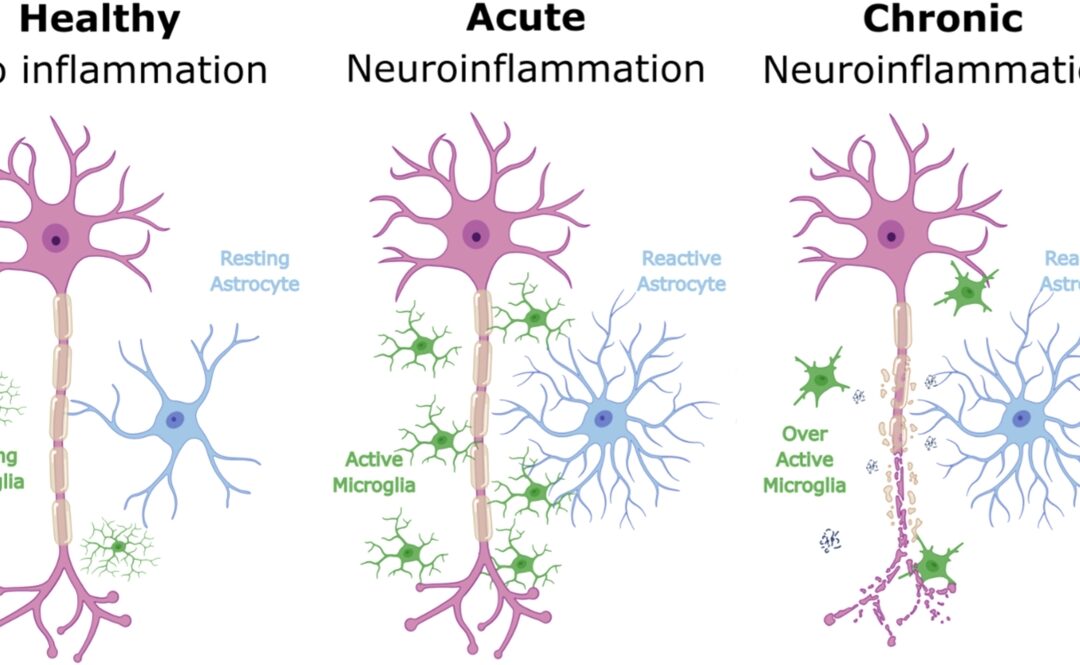
“Elevated CA-125 is typically associated with more aggressive disease biology and systemic inflammation. Because Elenagen reduces chronic inflammation, its effect may be most pronounced in patients where inflammation is stronger and/or is a primary driver of disease progression.”
He also pointed to antigen load as a possible factor.
“Patients with elevated CA-125 levels often reflect a larger load of tumor-associated antigens. By increasing tumor-infiltrating lymphocytes and reducing intratumoral immunosuppression, Elenagen may provide a more robust benefit for patients whose tumors demonstrate a higher antigen load.”
However, he underscored that these findings remain exploratory.
“The study was not powered to definitively establish predictive biomarker relationships; however, the signal certainly warrants prospective investigation.”
Such observations echo other areas of oncology, where patients with poor prognostic markers, such as BRCA mutations in certain cancers, may demonstrate heightened responsiveness to targeted or immune-based therapies.
Route of Administration: A Structural Advantage?
Beyond biology, Elenagen’s once-weekly intramuscular (IM) administration may carry meaningful implications for care delivery.
Dr Gabriel Levin highlighted that delivery logistics can significantly shape real-world access.
“A simple intramuscular injection may sound like a small logistical detail, but in global oncology it can be transformative.”
He contrasted IM administration with the infrastructure required for many IV immunotherapies.
“Most IV immunotherapies require infusion chairs, pharmacy mixing under sterile conditions, cold-chain logistics, trained infusion nurses, and monitoring capacity for infusion reactions. Those requirements are routine in major cancer centers, but they become real barriers in many regions.”
An IM-administered therapy, he explained, has a fundamentally different delivery profile.
“It can be administered in outpatient clinics with far less dependence on infusion suites, specialized equipment, or prolonged chair time. That opens the door to decentralizing care.”
Levin emphasised that simplified delivery does not eliminate the need for safety monitoring and appropriate protocols. However, if efficacy and safety are confirmed in larger trials, the route of administration could enable broader implementation across community oncology programs and lower-resource settings.
“If efficacy and safety are confirmed, the route of administration becomes a powerful enabler of global access, an innovation not just in biology, but in delivery.”
Looking Ahead
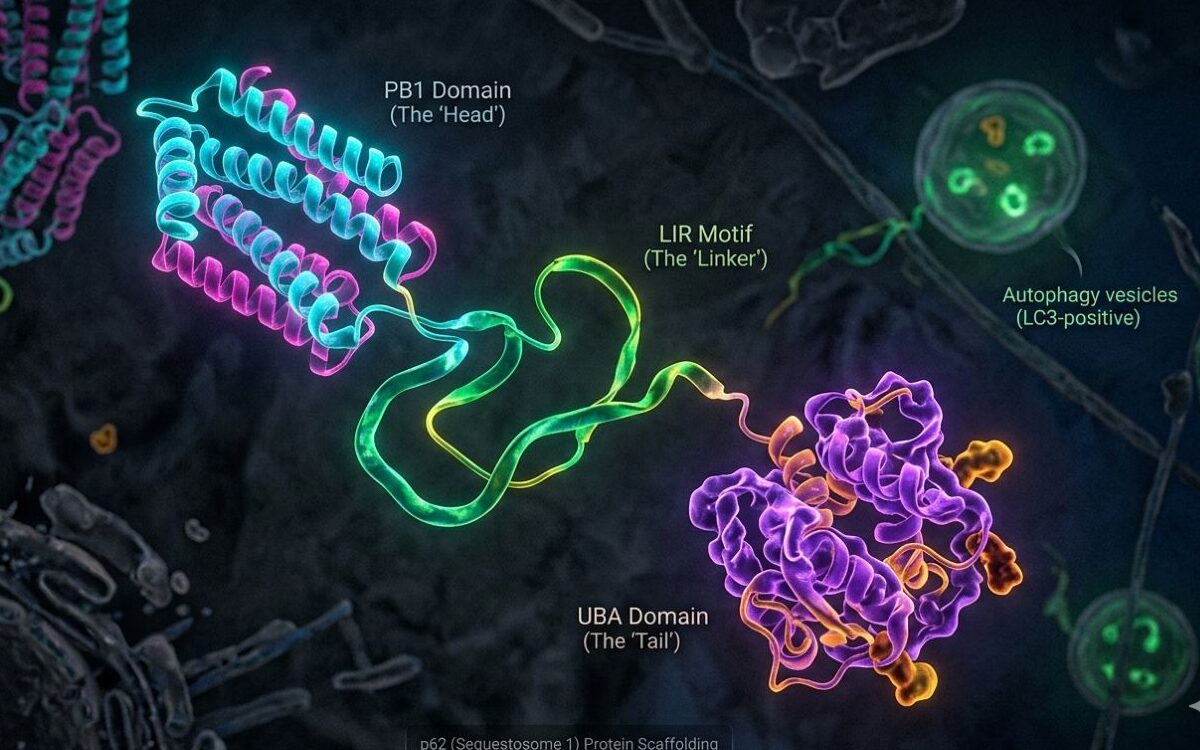
Elenagen is an investigational plasmid DNA therapy encoding the human protein p62/SQSTM1 and is designed to reduce chronic inflammation while reshaping the tumor microenvironment. The program is preparing for Phase II/III development in collaboration with the Gynecologic Oncology Group (GOG) Foundation, with additional exploration planned in aggressive breast cancer settings.
Platinum-resistant ovarian cancer remains an area of profound unmet need. While larger confirmatory trials will be required, the Phase II findings presented at ESGO 2026 introduce a combination of survival benefit, favorable tolerability and delivery practicality that is likely to prompt robust discussion within the oncology community.
As the field continues to explore immune modulation beyond checkpoint inhibition, Elenagen’s data may represent not just a clinical signal but a broader conversation about inflammation, immune architecture and how cancer therapies are delivered worldwide.
About Elenagen
Elenagen is an investigational DNA therapy consisting of a plasmid encoding the human protein p62/SQSTM1. In clinical studies conducted outside the U.S., Elenagen demonstrated a strong safety profile and statistically significant clinical benefit when combined with chemotherapy, along with evidence of immune activation and mitigation of chronic inflammation.
About CureLab Oncology
CureLab Oncology Inc. is a clinical-stage biotechnology company headquartered in the greater Boston area. The company is focused on developing safer, more effective treatments for solid tumors and inflammatory conditions. CureLab’s lead program, Elenagen, represents a novel DNA-based approach to cancer therapy. Learn more at curelaboncology.com.
Editorial Disclosure: This article is a contributor-led report based on clinical data, official press releases, and direct commentary provided by CureLab Oncology and Dr. Gabriel Levin. While the findings discussed were recently published in the International Journal of Gynecological Cancer, the specific insights and mechanistic explanations featured here represent the perspectives of the study’s investigators and the CureLab R&D team.