AI developments are happening at breakneck speed, with every sector and industry seeking new opportunities to seize and capitalise upon. AI-driven research is creating a fundamental shift for pharmaceutical competitive intelligence, moving away from a reactive, manual monitoring function to a predictive, real-time strategic engine.
Traditionally, competitive intelligence has relied on teams of analysts to comb through and piece together increasingly fragmented data sets. But today, AI platforms can automate insight extraction from channels like scientific literature, clinical trials, conference presentations, and company reports to anticipate and understand competitor moves months or even years in advance. With AI continuously monitoring and reporting on critical healthcare data sets, market trends, and competitor movements, corporate strategy and competitive intelligence teams at pharmaceutical corporations can focus their time on evaluating results and making confident decisions.
This transformation is creating a reengineered competitive playbook in which the most modern pharma companies are successfully deploying end-to-end AI workflows to outpace legacy organisations both in speed and transforming insight into action.
But what does this mean in practice?
The practical impact of AI on the drug development lifecycle
Perhaps the most meaningful competitive advantage for pharma companies using AI is the potential for a drastic reduction in research and development timelines. Pharmaceutical firms are getting ahead by adopting platforms with domain-specific data and content that provide a single, integrated view of the competitive landscape for greater efficiency and more accurate results versus juggling many different research tools.
For many, the ability to delve into clinical trial updates, government regulatory activity, disease landscaping, and partnership, merger, and acquisition deals in one unified space provides a decision-ready view of the market.
While AI research is redefining keeping pace with innovation, AI is also creating a material impact on the speed in which drugs are being developed.
An example is seen with preclinical acceleration; according to the UK’s “AI for science strategy”, one of the missions is to “accelerate drug discovery to develop trial-ready drugs within 100 days by 2030 and contribute to deploying new treatments faster.” AI also offers more efficient screening practices, with AI tools able to narrow down a library of 100 compounds to the most promising leads in one to two days, a process that typically takes a month using manual, brute force methods.
But what about the speed-accuracy trade off? For drug development, the early signals are positive, with AI-designed drugs showing an 80-90% success rate in safety trials, nearly double the historical average of 40% to 65%.
The significant reduction in timeline and increase in success rate is pivotal for drug development. Even marginal improvements in proficiency through AI can allow a company to surge ahead of its competitors, with firms using AI in research and development gaining the potential to understand a market need, analyse relevant factors and market signals, and bring a drug to market faster than a competitor.
The evolving intersection between AI, commercial strategy, and clinical decision-making
But speed isn’t the only benefit. AI is allowing commercial strategy and clinical decision-making to reposition as a unified, data-driven ecosystem where business operations and patient care are no longer operating separately.
Acting as the connecting point, AI is allowing pharmaceutical companies to re-evaluate and engage differently with the broader network. Life sciences companies are now empowered to use clinical insights to drive market strategy, all while providing clinicians with real-time, evidence-based tools to improve patient care.
The shift among pharma and biotech companies can be viewed as adapting from generic “one-size-fits-all” models to highly targeted, AI-enabled execution. Companies of every size in the industry are scaling AI across commercial operations toward better business outcomes like dynamically targeting their audiences, personalising messaging, and making more targeted investment decisions. For example, Pfizer is scaling AI across 1,200 GPUs specifically for dynamic targeting, research, and development. In addition, commercial teams are using AI to identify exact patient cohorts that are most likely to benefit from a therapy or drug treatment.
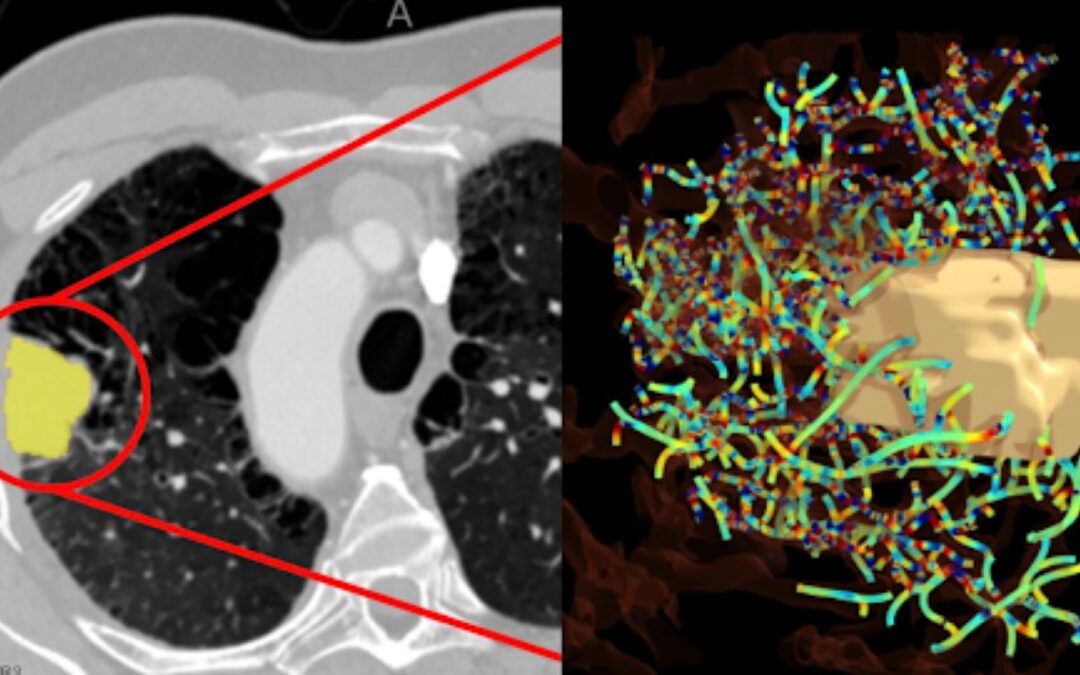
When it comes to actual clinical outcomes, AI is already transforming decision-making. Intelligent Decision Support Systems (IDSS) analyse vast datasets to flag potential drug interactions, prioritise high impact alerts, and reduce alert fatigue for clinicians. AI-powered imaging analysis also is proving reliable in diagnosing and detecting health concerns, with accurate results being reported within cancer detection.
The new battleground for AI-led pharma
In practice, the shift to AI-driven competitive intelligence isn’t just about speed or efficiency, but about who can make better decisions, faster, and more confidently. The real differentiator in pharma and biotech isn’t just using AI – it is between those that treat AI as a bolt-on efficiency tool compared to those that actually rebuild their decision making around it in areas such as go-to-market strategies, maximizing operational key performance indicators, and strengthening agility against competitors.
As pharma companies deepen their reliance on AI for competitive edge, transparency, validation, and human oversight must remain central. These fundamentals are critical to integrating AI successfully, and organizations that combine a human-centric approach with AI-led insight and domain expertise will achieve true competitive edge.