For most of the twentieth century, the global pharmaceutical industry operated on a straightforward geographic logic. Drugs were discovered and developed in North America and Western Europe, approved by the FDA and EMA, and then exported to the rest of the world. The rest of the world was a secondary consideration, a distribution challenge rather than a strategic opportunity. That model is now giving way to something considerably more complex, and considerably more interesting. The emergence of pharmerging markets, fast-growing developing economies now reshaping global drug development strategy, is central to understanding that shift.
The term “pharmerging” was coined by the market intelligence firm IQVIA to describe a group of approximately 21 high-growth developing economies whose pharmaceutical markets are expanding at rates that materially outpace those of mature Western markets. The category includes China, India, Brazil, Russia, Turkey, Mexico, and a cluster of Southeast Asian nations, and it has become one of the defining structural forces in global drug development strategy. According to analysis by Mordor Intelligence, the pharmerging market reached $2.18 trillion in 2025 and is forecast to reach $3.45 trillion by 2030, growing at a compound annual growth rate of 9.66%. These markets collectively represent more than 85% of the global population, and as their healthcare infrastructure, regulatory sophistication, and purchasing power continue to develop, they are no longer simply consumers of pharmaceutical innovation. Increasingly, they are helping to shape it.
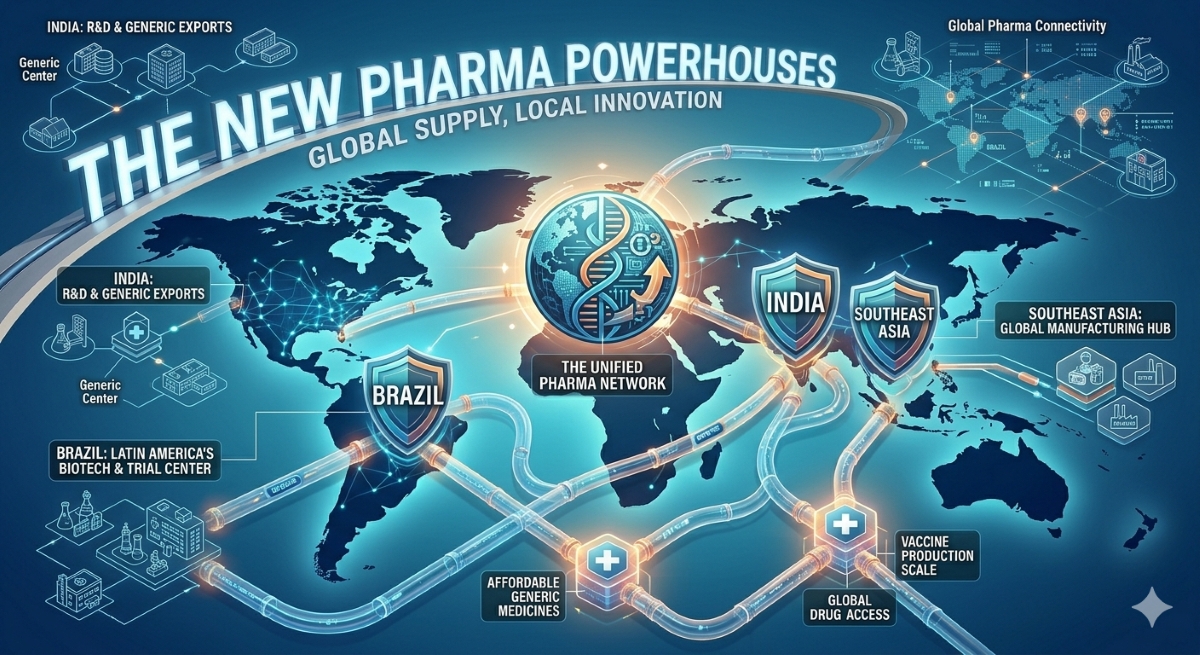
India: From Generic Powerhouse to Innovation Hub
India’s transformation in the global pharmaceutical value chain has been one of the most consequential shifts in the pharmerging markets story over the past two decades. The country is already the world’s largest supplier of generic medicines and a leading exporter of vaccines, with the United States, United Kingdom, and Brazil among its primary export destinations. But the strategic ambition being pursued by the Indian government and pharmaceutical industry now extends well beyond generics, a trajectory recently illustrated by India’s largest pharmaceutical company making its biggest-ever international acquisition.
The Production Linked Incentive scheme, launched in 2020 and expanded in subsequent years, has been the centrepiece of India’s pharmaceutical industrial policy. The scheme offers financial incentives to manufacturers of high-value pharmaceutical products including biopharmaceuticals, complex generics, patented and near-patent drugs, cell and gene therapies, orphan drugs, and active pharmaceutical ingredients. By March 2025, cumulative investment under the PLI Pharmaceuticals scheme had reached ₹37,306 crore, significantly exceeding the original target of ₹17,275 crore. Cumulative sales of approved products reached ₹2,66,528 crore, including exports of ₹1,70,807 crore, according to official data from India’s Department of Pharmaceuticals. Over the six financial years to 2024 to 2025, pharmaceutical exports grew by 92%, rising from ₹1,28,028 crore to ₹2,45,962 crore. India’s share of global bulk drug exports now stands at 30%.
The PLI scheme has also driven a structural shift in India’s API supply chain. For decades, India was heavily dependent on China for the bulk drugs that underpin its generic formulation industry, with import dependence estimated at around 72% for certain critical categories. Under the scheme, 726 APIs, key starting materials, and drug intermediates are now produced domestically, including 191 manufactured in India for the first time. The transition from net importer to net exporter of bulk drugs, achieved by 2024 to 2025, is a supply chain milestone that has not gone unnoticed by multinational pharmaceutical companies evaluating their procurement strategies in the context of ongoing geopolitical tensions.
India’s growing attractiveness as a clinical trial destination adds a further dimension to its pharmaceutical ascent. The country’s large and genetically diverse population, its concentration of English-speaking physicians trained to international GCP standards, and its relatively competitive site costs have historically made it an appealing location for Phase III trials. India is now becoming one of the largest clinical trial hubs in Asia, following China and Japan. The government’s Promoting Research and Innovation in Pharma-MedTech scheme, which established seven Centres of Excellence at National Institutes of Pharmaceutical Education and Research, is investing ₹700 crore in research infrastructure and industry-academia collaboration, with over 378 research papers and 33 patents filed in 2025 to 2026 alone.
The launch of Eli Lilly’s Mounjaro in India in late March 2025, in pre-filled injector pen form across six dose strengths, illustrated how rapidly the country’s market for innovative therapies is maturing. Chronic diseases including diabetes and obesity are rising sharply across India’s urban and semi-urban populations, and global manufacturers are increasingly developing market entry strategies that treat India as a priority launch market rather than an afterthought.
Brazil: Regulatory Reform and Clinical Trial Renaissance
Brazil presents a different but equally compelling story of pharmaceutical market transformation. As one of the core pharmerging markets, Brazil combines demographic scale with rapidly improving regulatory infrastructure. Latin America’s largest economy has long been recognised as a market with exceptional clinical trial potential, owing to its population of more than 212 million people, high disease prevalence across a range of therapeutic areas, and a network of research-capable hospital centres. The persistent barrier has been regulatory unpredictability. ANVISA, Brazil’s health surveillance agency established in 1999, developed a reputation for lengthy and inconsistent review timelines that made Brazil a lower priority in global clinical trial feasibility decisions despite its demographic advantages.
That is changing. The enactment of Law 14.874 introduced a framework that mandates strict review timelines, with ANVISA committed to a total turnaround time of 90 business days for responses following the submission of a new clinical study. For strategic public health products, this is reduced to 15 days. The removal of requirements for dual ethical approvals, previously a significant procedural burden, further streamlines the pathway to trial initiation. An ANVISA official, cited by pharmaboardroom.com in December 2025, stated that the new regulations position Brazil to attract additional clinical research across all trial phases, with particular opportunity in Phase I and II studies, where Brazil had historically been underrepresented.
Rogerio Frabetti of Biogen, which had approximately ten ongoing Phase II and III protocols in Brazil at the time of that assessment, described the opportunity in direct terms, highlighting the country’s population diversity, its network of centres receptive to innovation, and governmental incentives as factors capable of attracting substantially more clinical trial investment.
The access pathway for innovative medicines, however, remains a structural challenge. According to Renato Porto, President of MNC association Interfarma, ANVISA’s regulatory performance has improved substantially, with approval now typically taking around two to three years. But the time for a medicine to reach the population is closer to five years, with reimbursement negotiations and incorporation processes representing the real bottleneck. Almost half of drugs approved by ANVISA never reach the market, according to data cited by Brazil’s Regulatory Chamber of the Drug Market. For multinational companies, navigating the gap between regulatory approval and actual patient access remains one of the defining challenges of the Brazilian market.
In the broader picture, however, Brazil is unambiguously attracting attention. The biosimilar wave associated with the approaching patent cliff is of particular relevance: DrugPatentWatch analysis notes that patents for 117 blockbuster molecules are set to expire in Brazil by 2028, creating a substantial opportunity for domestic and international biosimilar developers. The country’s unified public health system, the SUS, has a long track record of making health technology assessment decisions that favour biosimilar adoption at scale when products are available and affordable.
Southeast Asia: Diverse Markets, Shared Momentum
Southeast Asia’s role in the global pharmaceutical value chain is growing rapidly but unevenly, reflecting the extraordinary diversity of the region. The Association of Southeast Asian Nations encompasses markets ranging from Singapore, a highly sophisticated biomedical research and manufacturing hub, to Indonesia, the world’s fourth-most-populous country, whose healthcare system is still in the process of achieving universal coverage.
Indonesia’s pharmaceutical market has been transformed by the expansion of Jaminan Kesehatan Nasional, the national health insurance programme described as the world’s largest single-payer system, which was approaching coverage of more than 90% of the Indonesian population by the end of 2023. The expansion of insured access to medicines has driven a step-change in pharmaceutical demand, particularly for generic and essential medicines, and the Indonesian government has responded by promoting domestic manufacturing to reduce import dependence. The country’s 2026 Halal mandate, which requires pharmaceutical products sold in Indonesia to comply with Islamic certification standards, is adding a layer of regulatory complexity for multinational manufacturers but is simultaneously stimulating investment in Halal-compliant manufacturing infrastructure.
Vietnam and Thailand are both emerging as clinical trial destinations, with improving regulatory frameworks, high-quality research hospital networks, and populations presenting disease profiles that are of direct relevance to drug development programmes targeting Asian patient populations. The growing recognition that many large-scale trials conducted exclusively in Western populations may not generate efficacy and safety data fully transferable to Asian patients is one of the forces driving pharmaceutical companies to develop regional clinical evidence packages.
Singapore remains the region’s most mature pharmaceutical and biomedical science hub, with a regulatory environment aligned closely to international standards and a reputation as a gateway market for companies seeking a credentialled entry point into the wider Southeast Asian region.
Asia-Pacific as a whole accounted for 39.61% of the global pharmerging market share in 2024, according to Mordor Intelligence, and is forecast to post a compound annual growth rate of 11.89% through 2030, the fastest of any region globally.
Multinational Strategies: From Export to Integration
The scale and trajectory of pharmerging markets is driving a fundamental strategic reorientation among the world’s largest pharmaceutical companies. The historical export model, in which a drug approved in the US or Europe was subsequently filed in emerging markets as a secondary commercial exercise, is giving way to what analysts at DrugPatentWatch have described as deeply integrated “poly-local” operations, in which product development, manufacturing, clinical evidence generation, and market access strategies are designed from the outset with pharmerging markets as primary rather than secondary considerations.
AstraZeneca and Sanofi have both adopted explicit localisation strategies in China, embedding R&D functions, manufacturing, and regulatory affairs teams within the country rather than managing the market from Western headquarters. The same logic is beginning to apply in India and Brazil, where the combination of market size, manufacturing capability, regulatory maturity, and clinical trial potential is compelling enough to justify the investment in genuinely local infrastructure.
Eli Lilly’s 2025 rollout of Mounjaro across India, China, Brazil, and Mexico, supported by $23 billion in global manufacturing capacity investment since 2020, exemplifies the new approach. The decision to launch simultaneously across multiple pharmerging markets, rather than treating them as a trailing second wave after the US and European launches, reflects a commercial and strategic calculus in which these markets are understood as essential to the drug’s global commercial profile from launch.
The Persistent Challenges
The growth story of pharmerging markets is genuine, but it is not unqualified. Intellectual property enforcement remains inconsistent across much of the region: analysis of China’s market found that 73.1% of drugs received approval before patent filing, and that 85.1% of generic applications lacked patent listings, creating conditions that discourage high-value R&D investment and fuel counterfeit proliferation. India’s price control framework has squeezed margins for smaller local manufacturers, with price caps on essential medicines constraining revenue even as demand grows.
Supply chain resilience is another persistent vulnerability. The COVID-19 pandemic exposed the degree to which global pharmaceutical supply chains depend on a small number of concentrated manufacturing locations, and the geopolitical tensions of the mid-2020s have kept supply chain diversification at the top of the pharmaceutical industry’s strategic agenda. Cold-chain logistics remain inadequate in parts of Southeast Asia and rural Brazil, limiting access to temperature-sensitive biologics and vaccines in the populations that often most need them.
Regulatory harmonisation across Southeast Asia remains incomplete, requiring companies to navigate multiple national approval pathways that do not yet share the degree of mutual recognition that exists between, say, the FDA, EMA, and TGA in Western markets.
A Structural Shift, Not a Trend
The rise of pharmerging markets is not a passing trend driven by a single favourable economic cycle. It reflects a structural realignment of global pharmaceutical activity towards the regions where the majority of the world’s population lives, where the burden of chronic disease is growing fastest, and where the combination of policy reform, manufacturing investment, and clinical trial capability is creating the infrastructure for sustained pharmaceutical development.
For drug developers, investors, and regulators, the implications are far-reaching. Trial populations will increasingly reflect the genetic diversity of Asian and Latin American patients. Manufacturing supply chains will be less concentrated in Europe and North America. Regulatory intelligence will need to span not just the FDA and EMA, but ANVISA, CDSCO, BPOM, and the ASEAN framework. The companies that are building those capabilities now are positioning themselves for the pharmaceutical landscape of the 2030s. Those that are not face the risk of being structurally disadvantaged in markets that will account for a growing share of global pharmaceutical value.