Oncology drug development is more mechanistically advanced than ever. Scientists and researchers design therapies to block immune evasion, disrupt tumor metabolism, and rewire the microenvironment. Yet a costly problem remains: many patients who should respond to these treatments don’t, and clinical developers and oncologists often don’t understand why until it is too late.
Existing biomarkers help to guide patients to the right treatments, but they do not fully capture an important component of the tumor microenvironment that these therapies must navigate to work: the tumor vascular network. Many patients who appear to be strong immunotherapy candidates by molecular criteria still fail to derive meaningful benefit. Abnormal tumor vasculature may contribute to that gap in some patients.
The Missing Dimension in Clinical Development: Vasculature
This abnormal architecture can impair drug delivery and immune cell trafficking and may contribute to resistance across multiple therapeutic classes.
Despite two decades of anti-angiogenic research, oncology investigators have lacked a practical and scalable way to quantify tumor vascular biology and identify which patients are most likely to benefit. Crucially, we now recognize that the vasculature is not just a target for specific anti-angiogenic drugs. Many other anti-cancer agents impact the vessel network as a downstream effect, even if it is not their primary mechanism. Measuring this impact is essential to understanding how any therapy is truly performing.
While specialized imaging exists in research settings, these protocols often sit outside routine clinical development workflows. We are developing precision medicines against a structural target we cannot see. That signal has been embedded in routine CT imaging all along, and we finally have the tools to measure it.
QVT: Measuring Tumor Vasculature from Routine Imaging
Standard CT scans, routinely collected for clinical care and clinical development of investigational therapies, contain detailed data on three-dimensional vascular architecture. Because these scans are already embedded in standard care and most trial protocols, measuring this vasculature can be incorporated without adding procedures, modifying imaging protocols, or placing additional demands on patients.
In a study published in the Journal for ImmunoTherapy of Cancer, we developed and validated the Quantitative Vessel Tortuosity (QVT) score in 682 patients with non-small cell lung cancer treated with standard-of-care immune checkpoint inhibitors.
This AI-derived imaging biomarker quantifies multiple features of vascular architecture, including tortuosity, branching patterns, and vessel volume, and integrates them into a single interpretable metric. Higher scores reflect a more disorganized vessel network, whereas lower scores are more consistent with vascular normalization. In this cohort, the QVT score was associated with survival independently of RECIST-based response assessment and tumor volume.
Much as ctDNA provides a dynamic window into tumor burden, the QVT score may provide a vascular counterpart—characterizing baseline vascular architecture and tracking its evolution on treatment.
Importantly, changes in QVT may be detectable as early as the first on-treatment scan, potentially offering a mechanistic readout before conventional imaging assessments diverge.
These findings represent promising early evidence and highlight the potential of QVT as an exploratory biomarker in this setting. Further validation across additional tumor types and treatment contexts will be needed to establish its broader utility.
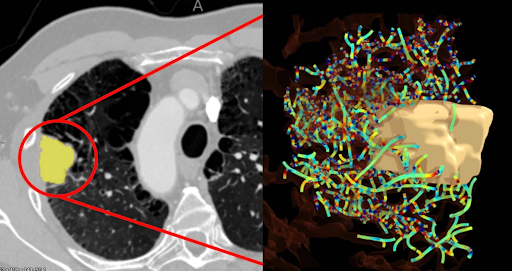
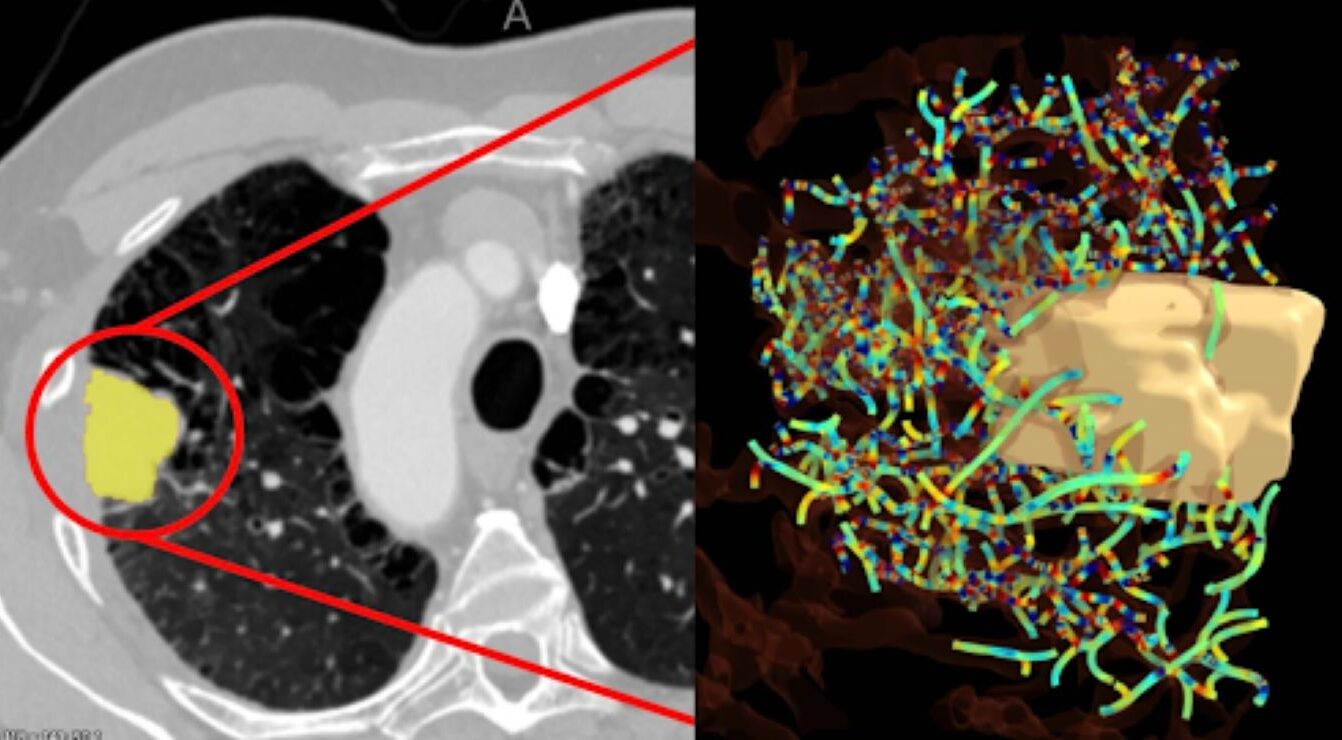
A routine CT scan of a lung tumor (left) is analyzed using Picture Health’s QVT™ score to map and quantify the complex network of blood vessels surrounding the tumor (right). Courtesy of Picture Health.
The Bispecific Frontier: A New Therapy Class
An important area in oncology drug development is the rapid rise of bispecific antibodies and combination strategies targeting both PD-(L)1 and VEGF. The biological rationale is compelling. Abnormal tumor vasculature does not only limit drug delivery; it actively suppresses immune function. Targeting VEGF therefore does double duty, improving the structural conditions for immune cell access while simultaneously relieving an active brake on the immune system. Paired with checkpoint inhibition, the combined effect is designed to be synergistic, not simply additive.
The clinical challenge is proving that the synergy is real. Overall survival remains the gold standard, but waiting years for survival data to mature creates a costly information gap during the most critical phases of development. This is particularly acute for bispecific programs, which are often evaluated against standard-of-care PD-1 therapies. Teams need to know earlier not just whether patients are responding, but whether the VEGF component is adding meaningful biological activity beyond checkpoint inhibition alone.
As a noninvasive imaging biomarker of vascular architecture, QVT may help address this gap. By assessing vascular structure at baseline and longitudinally on treatment, it has the potential to provide an early mechanistic signal of microenvironmental remodeling. In principle, this could give development teams additional evidence of biologic activity well before survival data mature, without adding burden to patients or requiring major protocol changes.
A Metric for the Next Decade of Development
For clinical investigators and drug development teams, the ability to quantify vascular architecture from routine imaging opens a new window into tumor biology. It offers a practical way to refine mechanistic understanding and strengthen decision-making during development.
Importantly, vascular imaging biomarkers can often be incorporated into trials using imaging that is already being collected. They may be applied retrospectively to existing datasets or prospectively in early-phase programs, adding a structural dimension to the evidence base with minimal incremental burden. As these tools continue to be developed and deployed in exploratory and research settings, further work will be needed to evaluate their performance across platforms, tumor types, and treatment settings before broad adoption.
For two decades, oncology has targeted tumor vasculature without a scalable way to measure it in routine development. That is beginning to change. As combination and bispecific strategies increasingly depend on vascular and immune biology, vascular imaging biomarkers deserve consideration as exploratory endpoints in trials already collecting serial CT imaging.
About the Author

Dr. Vamsidhar Velcheti MD, FACP, FCCP, FASCO is Professor of Medicine and Chief of Hematology and Oncology at Mayo Clinic Florida, where he holds the Florida Department of Health Chair of Cancer Research. His clinical and research focus spans thoracic malignancies, immune checkpoint therapy, and the development of AI-driven imaging biomarkers for oncology drug development. Dr. Velcheti is a co-author on the Journal for ImmunoTherapy of Cancer publication referenced in this article.
Disclaimer: Dr. Vamsidhar Velcheti is a co-author on the peer-reviewed publication referenced in this article. The QVT score described is a proprietary tool developed by Picture Health. This article represents the author’s independent expert commentary and does not constitute an endorsement by Life Science Daily News.