The emergence of GLP-1 receptor agonists has fundamentally reshaped obesity treatment. For the first time, we are seeing consistent, clinically meaningful weight loss at scale, supported by robust clinical programs such as STEP and SURMOUNT (Wilding et al., 2021; Jastreboff et al., 2022).
Yet as real-world experience expands, a more nuanced reality is becoming evident. A consistent pattern has emerged across both clinical practice and longitudinal studies: once therapy is discontinued, weight regain is not only common—it is often rapid and clinically significant (Rubino et al., 2021).
This is not a limitation of the drug, but a reflection of the disease itself. Obesity is chronic, adaptive, and biologically defended. When treatment is temporary, outcomes tend to be temporary as well.
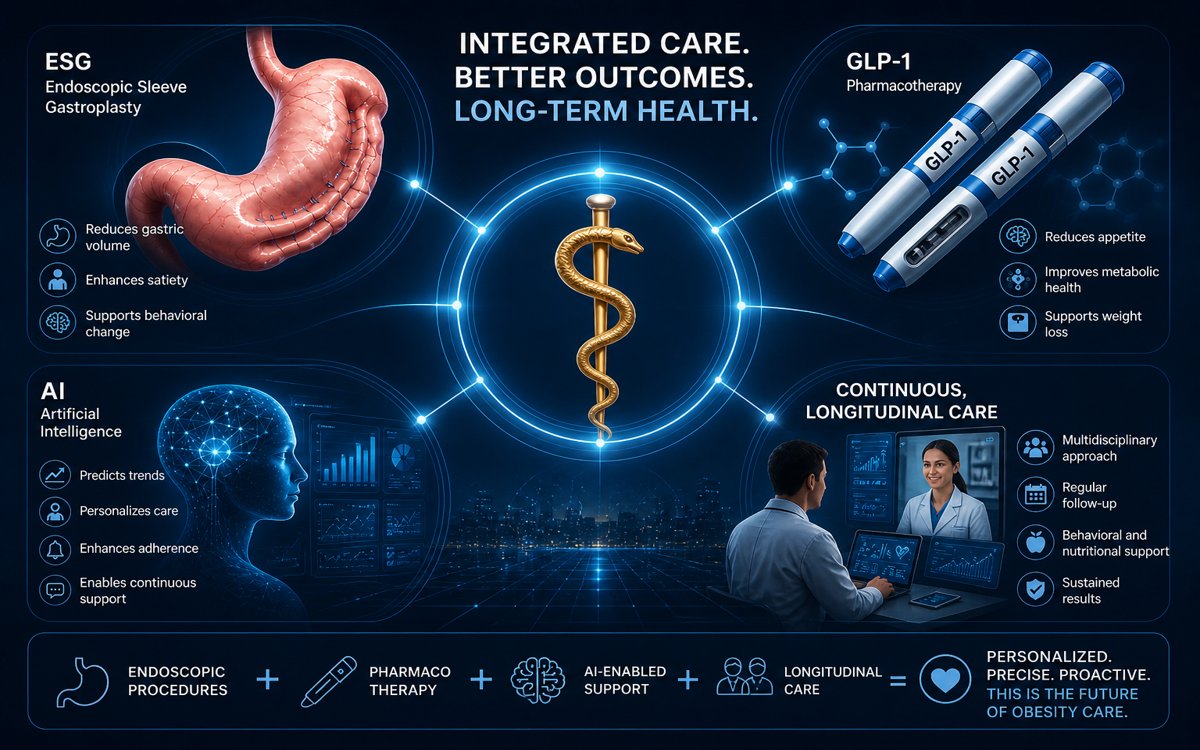
GLP-1 therapies are undeniably powerful tools. They modulate appetite, enhance satiety, and enable substantial weight reduction. However, they do not inherently reconfigure the behavioral, environmental, and structural drivers that sustain obesity over time.
In my clinical experience, patients who rely exclusively on pharmacotherapy often struggle with long-term maintenance once treatment is reduced or discontinued. Without a structured framework to support behavioral change, the underlying drivers of weight regain remain active.
This is where the limitation lies—not in pharmacologic efficacy, but in the model of care surrounding it.
Endoscopic therapies introduce a complementary dimension—one that is both physiological and behavioral. Procedures such as endoscopic sleeve gastroplasty (ESG) reduce gastric volume and alter gastric emptying dynamics, promoting earlier satiety and more structured intake patterns (Abu Dayyeh et al., 2015).
Their impact, however, goes beyond mechanics. They create a consistent physiological constraint paired with behavioral reinforcement, helping patients recalibrate eating patterns in a sustained and tangible way.
In my clinical practice, this often represents a turning point—not only in weight loss, but in how patients interact with food and engage with treatment over time.
Intragastric balloons offer an additional, complementary approach, particularly useful in initiating weight loss, enhancing early engagement, and bridging key phases of metabolic and behavioral transition.
The traditional dichotomy between pharmacological and procedural therapies is increasingly outdated. These modalities are not competing strategies, but synergistic tools within a broader therapeutic architecture.
When combined thoughtfully, they reinforce each other. Procedural interventions enhance satiety signaling and impose structural constraints, while pharmacotherapy stabilizes appetite regulation and mitigates metabolic adaptation.
In some cases, this synergy may allow for dose optimization of GLP-1 therapies while preserving clinical efficacy. More importantly, early and visible progress often strengthens patient motivation, improving adherence and long-term engagement.
Despite these advances, one of the most persistent limitations in obesity care is structural rather than therapeutic. Current models remain largely episodic, centered around isolated interventions—a prescription, a procedure, a follow-up visit—without a cohesive framework linking them over time.
This fragmented approach is fundamentally misaligned with the nature of a chronic disease.
Long-term success requires continuity: structured follow-up, early identification of disengagement, and integration of medical, nutritional, and behavioral care. Obesity management should not be event-based; it must be longitudinal and trajectory-driven.
As the field moves toward continuous care models, a practical challenge emerges: how to maintain this level of engagement at scale.
This is where artificial intelligence begins to play a meaningful role—not as a replacement for clinical judgment, but as an extension of the care team.
AI-supported systems can reduce administrative burden, streamline documentation, and facilitate continuous, low-friction patient interaction. They can help identify early behavioral drift, reinforce adherence, and maintain a sense of connection between visits.
In practice, this enables a model in which patients are not intermittently treated, but continuously supported.
In this context, artificial intelligence is less about automation and more about sustaining connection at scale.
Looking ahead, it is unlikely that any single therapy will define the future of obesity treatment.
Progress will come from integration—combining endoscopic interventions such as ESG and intragastric balloons with pharmacological therapies, multidisciplinary care, and structured long-term follow-up.
This approach acknowledges the full complexity of obesity, addressing not only biological mechanisms but also behavioral patterns and environmental influences that shape long-term outcomes.
GLP-1 therapies have undeniably transformed the landscape of obesity care. They have redefined expectations and expanded what is clinically achievable.
But they are not, in isolation, a complete solution.
Long-term success will depend on how effectively we integrate procedural, pharmacological, and behavioral strategies within a continuous model of care.
Because ultimately, obesity is not a condition that can be treated in isolated moments—it is one that must be managed over time.
Author Bio

Flavio Mitidieri Ramos, MD, MSc, FASGE
Dr. Flavio Mitidieri Ramos, MD, MSc, FASGE, is a gastroenterologist and advanced endoscopist with more than 18 years of clinical experience in therapeutic endoscopy and metabolic interventions. He is a Fellow of the American Society for Gastrointestinal Endoscopy (FASGE), a clinical researcher, and a peer reviewer for Springer Nature (Obesity Surgery) and Frontiers.
Dr. Ramos serves on international editorial boards and plays an active role in advancing minimally invasive bariatric therapies, including contributions to regulatory and CE mark renewal processes for gastric balloon technologies. He has served as a medical advisor to industry and as a consultant for medical technology companies, including Boston Scientific. He is among early adopters of bariatric endoscopy techniques in Brazil. His work focuses on integrating endoscopic, pharmacologic, and AI-enabled care models to optimize long-term obesity outcomes and improve patient adherence at scale.